Nomenclature of Alkenes
Alkenes are hydrocarbons that have double bond(s). In the naming of a hydrocarbon, you
use the suffix -ene. For instance, a straight chaine of six carbons would be called
hexene.
When naming alkenes, you must designate where the double bond occurs. Numbering occurs
on the lower numbered carbon.
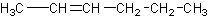
This would be 2-hexene. NOT 3-hexene. 3-hexene would be:
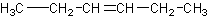
This is pretty easy, until you start getting into substituents.
Naming substituents in a straight chain (also called an n- chain) is pretty
basis. Name this compound:
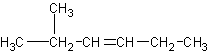
This would be called 2-methyl-3-hexene.
In organic, there is a whole "priority" in numbering. If you have a methyl-
group and a -ene group, then the -ene group gets priority (meaning that the -ene must the
lowest possible). For instance let's look at this molecule:
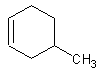
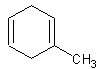
This would be called 4-methylcyclohexene. The -ene group has priority over the methyl-
group, so the -ene is assumed to be one. Then, counting in a counterclockwise direction
(clockwise would yield 5-methylcyclohexene, and lower numbers are preferred), the methyl
group is located on the 4 carbon.
When you have more than one -ene, you add a prefix to the suffix. For instance, if you
have TWO double bonds, the molecule would be -diene. Three double bonds would be called
-triene. Easy shtuff. Let's try some examples:
Well, the -ene gets priority from the methyl-group,and we also know there are two
double bonds, so we know to name this a methylcyclohexadiene. Now, in terms of numbering,
we can either have 4-methyl-1,4-cyclohexadiene or 1-methyl-1,4-cyclohexadiene. Since we
always want lower numbers, we choose the latter. Hence, this molecule is called
1-methyl-1,4-cyclohexadiene.

2,5-heptadiene. Easy.

Ignore the methyl for a second. We know that going both ways, it will be 2,5-heptadiene
(try counting carbons from the left and then from the right). So, we'll name the left
carbon "one" since the methyl group would then be 2-methyl as opposed to
6-methyl. Final answer? 2-methyl-2,5-heptadiene.

This is exactly like the last one, except we have one more double bond. I used this one
to demonstrate the priority of the double bond over the methyl- substituent. This one has
three double bonds, so it's a triene. It has 7 carbons, so it's a hepta-. However, this
one is NOT named the same way going from the right or the left (unlike the last problem).
Going from the right, this is a 1,2,5-heptatriene. Going from the left would be
2,5,6-heptatriene. So we MUST name from the right since it has lower numbering. Although
the methyl- group will be a 6-methyl-, the -enes draw the priority. Sort of like the
"pecking" order at a school. Teachers over students :)
Final answer? 6-methyl-1,2,5-heptatriene.