Cyclic alkanes and conformationsNaming cyclic alkanes
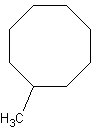
First you would count the longest carbon chain. You can see that the "cyclic"
ring has 8 carbons, so it's octane. Since it's a cyclic ring, you call it a
"cyclo" alkane. Hence, the main part of the molecule would be called
cyclooctane. The substituent is called a methyl group, so you get the name
methylcyclooctane.
Now, the hard part of this is figuring out numberings. You can pick ANY carbon to be
"1," so we'll conveniently choose the carbon that the methyl is on as
methylcyclooctane. It wouldn't matter if the methyl group was on the top right carbon,
because it would still be methylcyclooctane, because you could rotate that molecule to get
the one above. Now, let's try two substituents.
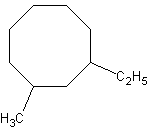
We again know it's a cyclooctane, but the problem arises in how to number the
substituents. We could name this 1-methyl-3-ethylcyclooctane, or
1-ethyl-3-methylcyclooctane. We name it 1-ethyl-3-methylcyclooctane because of the
alphabetization rules. Also, priority is given to the longest carbon chain substituent in
naming (so we wouldn't name it 3-ethyl-1-methylcyclooctane).
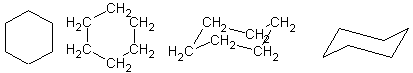
These are some ways to draw the same compound, cyclohexane. Cyclohexane is a pretty
important molecule, and I guarantee you will see these again.
Basically the last two are cyclohexane drawn the way it really does look. The 2nd
drawing is for "watered down" organic that is taught to AP Chemistry students.
If you take a real orgo class (although I've only taken an intro course) you will learn
all sorts of cool stuff like that.
Now, with the 4th cyclohexane, there are two positions for atoms.
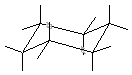
Note that this is the same molecule as the top right cyclohexane, except I've drawn
in all the hydrogens.
The lines going up and down are considered the "axial" position, while the
ones going right and left are considered "equatorial." Now, guess which position
is more stable. The equatorial is! If you have an axial position, there is steric
repulsion between all the axial atoms.