U.S. National Chemistry Olympiad: 1986 Local
Section Test
Go to the answers
1. A strip of silver foil has a uniform width of 1.00 cm and a uniform thickness of
0.0119 cm. If the density of silver is 10.5 g/cm3, how long a strip should be
cut to obtain 1.00 g of the metal?
(A) 9.52 x 10Ż2 cm
(B) 1.19 cm
(C) 8.00 cm
(D) 12.5 cm
2. A car traveling at 10 miles per hour emits about 0.15 kg of carbon monoxide (CO) gas
per mile. How many moles of CO are emitted per mile under theses conditions?
(A) 1.5 x 10Ż1
(B) 5.4
(C) 9.4
(D) 12.5
3. You need to fertilize an area with the equivalent of 25.0 pounds of nitrogen, N. How
much urea, CON2H4, should be used?
(A) 46.7 lb
(B) 53.6 lb
(C) 107 lb
(D) 127 lb
4. A sample of a conpound contains 0.100 g of hydrogen and 4.20 g of nitrogen. The
simplest formula for the compound is
(A) HN2
(B) HN3
(C) NH3
(D) NH2
5. If the hydrocarbon C2H4 is burned in oxygen gas, carbon
dioxide and water are formed as described by the unbalanced chemical equation
C2H4 + O2 ----> CO2 + H2O
When this equation is balanced properly, we predict that one mole of C2H4
will
(A) react with one mole of O2
(B) form two moles of CO2.
(C) form three moles of water.
(D) react with four moles of O2.
6. A human patient suffering from a duodenal ulcer may show a hydrochloric acid
concentration of 0.080 mol/liter in his gastric juice. It is possible to neutralize this
acid with aluminum hydroxide, Al(OH)3, which reacts with HCl according to the
balanced chemical equation
Al(OH)3 + 3 HCl ----> AlCl3 + 3 H2O
If the patient's stomach receives 3.0 liters of gastric juice per day, how much
aluminum hydroxide must he consume per day to counteract the acid?
(A) 6.2 g
(B) 19 g
(C) 26 g
(D) 78 g
7. The correct molecular formula of a compound that has an empirical formula of C2H2O
and a molecular weight of 168 is
(A) C2HO
(B) C4H24O6
(C) C8H8O4
(D) C8H12O5
8. Given the reaction
Na2O(s) + H2O(l) -----> 2 NaOH(aq)
what is the molarity of the solution formed if 1.35 g of Na2O is mixed with
water such that the final volume is 100 mL?
(A) 0.0435 M
(B) 0.108 M
(C) 0.217 M
(D) 0.435 M
9. When ignited in air, magnesium burns with a bright flame to form magnesium oxide.
The standard heat of formation of solid MgO is -1203 kJ/mol. How much heat is evolved when
sufficient magnesium meal is burned in oxygen to form 1.0 g of MgO?
(A) 0.025 kJ
(B) 30 kJ
(C) 40 kJ
(D) 1203 kJ
10. 40Ca, 39K, and 41Sc all have the same
(A) number of electrons.
(B) atomic number.
(C) mass number.
(D) number of neutrons.
11. 1s22s22p63s23p63d34s2
is the ground state electonic configuration of
(A) Ca
(B) Sc
(C) Zn
(D) V
12. The first ionization energy for a mole of magnesium atoms is the energy required
for the process
(A) Mg(s) ----> Mg(g)
(B) Mg(g) ----> Mg+(g) + 2eŻ
(C) Mg+(g) -----> Mg2+(g) + eŻ
(D) Mg(g) ------> Mg+(g) + eŻ
13. Element x, whose atoms have an outer-shell electron configuration ns2np4,
is most likely to react chemically to form ions which have a charge of
(A) +2e
(B) +e
(C) -e
(D) -2e
14. The formula of the compound ammonium carbonate is
(A) NH4CO3
(B) NH4CO4
(C) NH4HCO3
(D) (NH4)2CO3
15. Which statement regarding the properties of elements arranged in the Periodic Table
is correct?
(A) Atomic sizes decrease going down a Group or Family.
(B) Atomic sizes increase going from Fr in Group I to F in Group VII.
(C) Atomic sizes decrease going from left to right in a Period.
(D) All atoms in the same Group have the same size.
16. In their stable compounds, alkaline earth metals normally exist as
(A) M+ cations
(B) M2+ cations
(C) M atoms
(D) MŻ anions
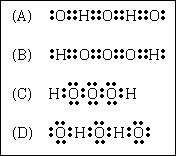
17. Hydrogen trioxide, H2O3, a highly unstable and reactive
compound related to hydrogen peroxide, has been prepared recently in small amounts. Which
Lewis electron dot structure is most likely to represent the structure of hydrogen
trioxide?
18. The most likely molecule to be formed by the reaction of boron and chlorine is
(A) BCl
(B) BCl3
(C) BCl4
(D) BCl6
19. In which compound would the molecules be expected to be linear?
(A) O3
(B) SO2
(C) H2O
(D) CO2
20. The shortest bond is that in
(A) F2
(B) O2
(C) N2
(D) C2
21. Which is the most electronegative atom?
(A) P
(B) Al
(C) Cl
(D) S
22. The closest verbal description of the shape of the NH3 molecule (defined
by the positions of the atomic nuclei) is
(A) tetrahedral
(B) non-linear
(C) trigonal planar
(D) trigonal pyramid
23. Which of these molecules is octahedral?
(A) SF4
(B) SF6
(C) ClF3
(D) BrF5
24. Given the reactions
| CH4(g) + 2O2(g) -----> CH2O (g) + 2H2O(g)
|
DH° = -890.4 kJ |
| CH2O(g) + O2(g) -----> CO2(g) + H2O(g) |
DH° = -563.5 kJ |
what is DH° for the reaction
CH4(g) + O2(g) ----> CO2(g) + H2O(g)?
(A) -1453.9 kJ
(B) -326.9 kJ
(C) -236.6 kJ
(D) +118.3 kJ
25. 500 mL of a gaseous compound weighs 0.9825 gram at 0 °C and 760 torr. What is the
approximate molecular weight of the compound in g/mol?
(A) 19.7
(B) 38. 7
(C) 44.0
(D) 58.9
26. What is the number of molecules in 1.00 mL of an ideal gas at STP?
(A) 2.69 x 1022
(B) 6.02 x 1020
(C) 2.69 x 1019
(D) 22.4 x 1019
27. If 6.60 g of a gaseous compound occupy a volume of 1.20 L at 27 °C and 0.967 atm,
the molecular weight of the compound is
(A) 109 g/mol.
(B) 123 g/mol.
(C) 140 g/mol.
(D) 165 g/mol.
28. How many liters of hydrogen gas at STP can be produced by the reactionof 9.00 g of
Al with excess dilute H2SO4?
2 Al(s) + 3 H2SO4(aq) -----> 3 H2(g)
+ Al2(SO4)3
(A) 5.61 L
(B) 11.2 L
(C) 33.6 L
(D) 67.2 L
29. Which substance has the highest boiling point?
(A) CH4
(B) He
(C) HF
(D) Cl2
30. When sugar is dissolved in water, the resulting solution
(A) has a higher freezing point than pure water.
(B) has a higher boilng point than pure water.
(C) has a higher vapor pressure than pure water.
(D) has a lower osmotic pressure than pure water.
31. The process of dissolving solid ammonium nitrate in water is endothermic. Which
statement is true?
(A) The solubility of ammonium nitrate becomes larger at higher temperatures.
(B) The solubility of ammonium nitrate becomes lower at higher temperatures.
(C) The solubility of ammonium nitrate is not affected by temperature.
(D) One cannot predict the solubility behavior of ammonium nitrate from the information
given.
32. A compound containing an oxygen atom in the +2 oxidation state is
(A) OF2
(B) H2O
(C) K2O2
(D) Li2O
33. In the reaction
6 KI + 2 KMnO4 + 4 H2O ------> 3 I2
+ 2 MnO2 + 8 KOH
which atom undergoes oxidation?
(A) K
(B) Mn
(C) O
(D) I
34. For the unbalanced oxidation-reduction reaction
Ag2S(s) + NO3Ż (aq) ----> Ag+ (aq)
+ S(s) + NO(g)
which takes place in an acidic solution, what is the balanced half-reaction for the
process that involves reduction?
(A) Ag2S(s) ---> 2 Ag+(aq) + S(s) + NO(g)
(B) S2Ż(aq) ---> S(s) + 2eŻ
(C) NO3Ż (aq) + 4 H+(aq) + 3eŻ ---> NO(g) + 2 H2O(l)
(D) NO3Ż (aq) + 2 H+(aq) + 2eŻ ---> NO(g) + 2 OHŻ(aq)
35. In the Arrhenius picture of acids and bases, the reaction of an acid with a base
always results in the formation of
(A) H+
(B) OHŻ
(C) CO2
(D) H2O
36. A basic oxide is
(A) CaO
(B) N2O5
(C) CO2
(D) SiO2
37. A standard solution of 0.165 M HCl is being used to determine the concentration of
an unknown NaOH solution. If 25.5 mL of the acid solution are required to neutralize 15.0
mL of the base, what is the molarity of the NaOH solution?
(A) (0.165) / (25.5 + 15.0) M
(B) (15.0) / (0.165) (25.5) M
(C) (0.165) (15.0 / 25.5) M
(D) (0.165) (25.5 / 15.0) M
38. The half-life of radioactive 55Cr is 1.8 hours. The delivery of a sample
of this isotope from the reactor to your laboratory requires about 10.8 hours. What is the
minimum amount of such material that should be shipped in order that you receive 1.0
milligram of 55Cr?
(A) 128 mg
(B) 64 mg
(C) 32 mg
(D) 11 mg
39. A catalyst that increases the rate of a reaction does so by
(A) increasing the concentrations of the initial reactants
(B) increasing the temperature
(C) decreasing the temperature
(D) decreasing the activation energy for the process
40. Seawater contains a fairly high concentration of Mg2+(aq) ion, about
0.05 M. Seawater therefore would not be expected to contain very large concentrations of
(A) ClŻ
(B) SO42Ż
(C) K+
(D) OHŻ
41. 500 mL of 0.020 M BaCl2(aq) is added to 500 mL of 0.020 M NaF(aq). Ksp
= 2 x 10Ż6 for BaF2 in water.
(A) The solutions mix without reaction
(B) A precipitate of BaF2(s) is formed when the solutions are mixed
(C) Undissolved BaCl2 solid dissolves when the solutions are mixed
(D) The concentrations of all the ions in the final solution are 2 x 10Ż6 M.
42. Which aqueous solution should be neutral?
(A) NH4Cl
(B) Na(ClO4)2
(C) KCN
(D) NaHSO4
43. The pH of a solution of vinegar is 3.00. The concentration of OHŻ ion in this
solution is
(A) 3.00 M
(B) 1 x 10Ż3 M
(C) 1 x 10Ż11 M
(D) 17 M
44. The indicator ravishing red has a color change from yellow to red in the pH range
6.5- 7.5. This indicator would be used to titrate
(A) a weak acid with a strong base
(B) a strong acid with a weak base
(C) a weak acid with a weaker acid
(D) a strong acid with a strong base
45. If the Ka of a weak acid is 5 x 10Ż4, the pH of a 0.2 M
solution of the acid will be about
(A) 0.7
(B) 2.0
(C) 3.3
(D) 7.0
46. Given the standard electrode (reduction) potentials:
| Cd2+(aq) + 2eŻ ---> Cd(s) |
E° = -0.40 v |
| Ag+(aq) + eŻ ---> Ag(s) |
E° = +0.80 v |
What would be the E° for a cadmium-silver cell?
(A) 0.4 v
(B) 0.5 v
(C) 1.2 v
(D) 2.0 v
47. Consider the following reaction at equilibrium in a container of
constant volume:
| 2 SO2(g) + O2(g) <===> 2 SO3(g) |
DH = -7.8 kcal |
Which of the following would result in a greater concentration of SO2?
(A) addition of O2
(B) addition of SO3
(C) removal of SO3
(D) a decrease in temperature
48. A current of 10.0 amperes flows for 2.00 hours through an electrolytic cell
containing a molten salt of metal x. This results in the decomposition of 0.250 mole of
metal x at the cathode. The oxidation state of x in the molten salt is
(A) 1+
(B) 2+
(C) 3+
(D) 4+
49. In a voltaic cell, oxidation occurs at the
(A) anode
(B) cathode
(C) salt bridge
(D) electrode at which electrons enter from the outside
50. The free energy change for the chemical reaction that occurs in a voltaic cell when
it is discharging and producing an electric current must be
(A) positive
(B) negative
(C) zero
(D) unpredictable
51. The average distance of an electron in a hydrogen atom from the nucleus is
indicated by the quantum number
(A) n
(B) ml
(C) l
(D) ms
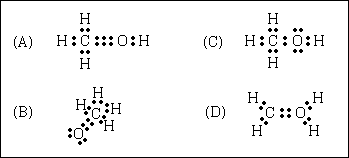
52. "Wood alcohol" (methyl alcohol or methanol), CH4O, is toxic
and is used as a denaturant in industrial grain alcohol and as a fuel. A satisfactory
Lewis structure for this compound is
53. In the SiH4 molecule, the Si atomic orbitals used in forming covalent
bonds are best described as
(A) one s and three p atomic orbitals
(B) four sp3 hybrid orbitals
(C) four d2sp3 hybrid orbitals
(D) three sp2 orbitals and one p orbital
54. The triple bond in the N2 molecule can be described as
(A) two bonding and one antibonding sigma bonds
(B) three sigma bonds
(C) one sigma and two pi bonds
(D) two bonding and one antibonding pi bonds
55. Which one of the following is not the expected geometry of the indicated molecule
or ion?
(A) CCl4 - regular tetrahedron
(B) BeCl2 - linear
(C) SO3 - triangular pyramid
(D) CH2O - trigonal planar
56. A 0.10 molal aqueous solution of HF shows a freezing point of -0.198 °C. What is
the percent dissociation of HF into ions in this solution? (Kf = 1.86 for
water)
(A) 6.4%
(B) 10%
(C) 20%
(D) 98%
57. Which one of these solid substances has a crystal structure containing separate
discrete molecules?
(A) Dry ice
(B) Graphite
(C) Quartz
(D) Silver bromide
58. A solution of a non-electrolyte, x, contains 84 grams of x per kilogram of water
and freezes at -1.46°C. What is the molecular weight of x? (Kf = 1.86)
(A) 84 x 1.86 x 1.46 = 222 g/mol
(B) 84 x (1.86 / 1.46) = 107 g/mol
(C) 84 x (1.46 / 1.86) = 66 g/mol
(D) 1.46 x (1.86 / 84) = 0.032 g/mol
59. A 1.20 liter sample is drawn from a bottle labeled "86.0% by weight H2SO4,
density 1.787 g/mL". What is the molarity of the sample?
(A) 8.78 M
(B) 10.5 M
(C) 15.7 M
(D) 18.2 M
60. Which substance is most soluble in water?
(A) C6H6
(B) CH3OH
(C) CO2
(D) CH4
61. 2.80 grams of a monoprotic weak acid, HX, was dissolved in water. Titration of the
acid to its equivalence point required 29.2 mL of 0.500 M NaOH solution. What is the
molecular weight of the acid, HX?
(A) 192 g/mol
(B) 164 g/mol
(C) 96.0 g/mol
(D) 5.21 g/mol
62. The ionic reaction
3 IŻ + S2O82Ż ---> 2 SO42Ż
+ I3Ż
is found to be described by the experimental rate equation
rate = k[IŻ] [S2O82Ż]
This suggests that
(A) the reaction occurs when three iodide ions collide simultaneous with one S2O82Ż
ion
(B) there must be only one step in the overall reaction since the rate is first-order in
both reactants
(C) the rate of reaction is inhibited by the presence of I3Ż
(D) a slow step in the overall reaction sequence may involve the collision of a single IŻ
ion with a single S2O82Ż ion
63. The hemoglobin from the red corpuscles of most mammals contains approximately 0.33%
iron by weight. If physical measurements indicate a molecular weight for hemoglobin of
68000 g/mol, how many iron atoms are there in each molecule of hemoglobin? (AW Fe= 56)
(A) 1
(B) 2
(C) 3
(D) 4
64. Given the following standard enthalpies of formation in kJ/mol:
CO2 -393.3, CH3OH -238.5, H2O -285.3.
What is the heat of combustion of methanol, CH3OH, in kJ/mol?
(A) -725.4
(B) -154.8
(C) -140.8
(D) 154.8
65. An element whose neutral atoms would be expected to be paramagnetic is
(A) neon
(B) fluorine
(C) magnesium
(D) zinc
66. The heat of formation of gaseous water molecules is -241.8 kJ/mol. If the H-O bond
energy is 463 kJ/mol and the H-H bond energy is 435 kJ/mol, what is the energy of the bond
between oxygen atoms in the O2 molecule in kJ/mol?
(A) 0
(B) 249
(C) 428
(D) 498
67. The osmotic pressure of an aqueous solution containing 30.0 g of a newly-isolated
protein in one liter of solution is 12.7 torr at 25 °C. What is the approximate molecular
weight of the protein in g/mol?
(A) 58
(B) 333
(C) 4000
(D) 44000
68. Carbon dioxide is a gas at room temperature while silicon dioxide is a high-melting
solid. The best explanation of this difference is that
(A) Si has more electrons than C
(B) the atoms in CO2 are bound by covalent bonds while SiO2 is an
ionic compound
(C) van der Waals' forces are stronger in SiO2
(D) CO2 consists of discrete molecules while in SiO2 atoms are
joined by a network of covalent bonds
69. 2.00 moles of NO and an undetermined amount of O2 are placed in a
one-liter container at 460 °C. When the reaction
2 NO(g) + O2(g) <===> 2 NO2(g)
reaches equilibrium, we find 0.00156 mol of O2 and 0.500 mol of NO2.
The value of the equilibrium constant for this system is
(A) 4.42
(B) 40.1
(C) 71.2
(D) 214
70. Proteins are macromolecules in which the simplest repeating structural units are
(A) nucleic acids
(B) amino acids
(C) purines
(D) carbohydrates
|