U.S. National Chemistry Olympiad: 1990 National
Test
Go to the answers
1. In moving from left to right across a period in the periodic table of the elements
(A) ionization energy decreases
(B) atomic radius decreases
(C) electronegativity decreases
(D) electron affinity decreases
2. Which transition is associated with the largest change in energy in the hydrogen
atom?
(A) n = 5 to n = 3
(B) n = 2 to n = 1
(C) n = 3 to n = 2
(D) n = 4 to n = 2
3. In which pair are the elements most similar in their chemical properties?
(A) B and N
(B) Li and Fr
(C) Mg and Al
(D) S and Cl
4. A 4.0 g sample of impure Ca(NO3)2 was found to contain 0.85 g
of calcium. What percentage of Ca(NO3)2 was in the original sample?
(molar mass of Ca(NO3)2 = 164.1 g mol�1)
(A) 13 %
(B) 36 %
(C) 64 %
(D) 87 %
5. Aluminum hydroxide, Al(OH)3, is insoluble in water, but dissolves readily
in both acidic and basic solutions. Such behavior is characteristic of
(A) polyprotic behavior.
(B) hydrophilic behavior.
(C) amphoteric behavior.
(D) a buffer.
6. The freezing point of a 1.00 m aqueous solution of HF is found to be -1.91
�C. The freezing point constant is water, kf, is 1.86 K m�1.
What is the percent of dissociation of HF at this concentration?
(A) 2.7 %
(B) 5.2 %
(C) 10 %
(D) 30 %
7. Enthapies of formation cannot be measured for many compounds
directly, and must be calculated from combustion data. Given the following data, calculate
the heat of formation of glucose, C6H12O6.
| C (graphite) + O2 (g) --> CO2 (g) |
DH = -394 kJ mol�1 |
| H2 (g) + 1/2 O2 (g) --> H2O (l) |
DH = -286 kJ mol�1 |
| C6H12O6 (s) + 6 O2 (g) --> 6 CO2
(g) + 6 H2O (l) |
DH = -2808 kJ mol�1 |
(A) -1272 kJ mol�1
(B) +1272 kJ mol�1
(C) +2128 kJ mol�1
(D) -3488 kJ mol�1
8. If the enthalpy of fusion of iodine is +15.5 kJ mol�1, and the enthalpy
of sublimaiton of iodine is +57.3 kJ mol�1, what is the enthalpy of
vaporization of iodine?
(A) -41.8 kJ mol�1
(B) +41.8 kJ mol�1
(C) -72.8 kJ mol�1
(D) +72.8 kJ mol�1
9. If, for N2, DHvap = 5.58 kJ mol�1
and DSvap = 72.1 J mol�1 K�1,
at low temperatures, in what phase will N2 be at -165 �C?
(A) gas
(B) liquid
(C) solid
(D) at the liquid/gas phase boundary
10. Which of the following molecules contains a central atom which is sp2
hybridized?
(A) H2SO4
(B) H2CO3
(C) ICl2
(D) H3CCH3
11. Which of the following molecules has at least one non-bonding pair of electrons on
the central atom?
(A) CHCl3
(B) HCN
(C) H2CO
(D) O3
12. Which of the following statements about ionic compounds is false?
(A) Ionic compounds are hard, brittle solids with high melting points.
(B) Some compounds which exhibit primarily ionic bonding in the solid phase can form
covalent bonds in the gas phase.
(C) The lattice energy is the quantity of energy required for 1 mol of a solid ionic
substance to be separated completely into gaseous ions well removed from one another.
(D) In general the lattice energy increases as the charge on the anion and cation increase
and increases as the size of the anion and cation decrease.
13. The pH of a saturated solution of calcium hydroxide is 12.40. What is Ksp
for this salt?
(A) 6.3 x 10�38
(B) 1.58 x 10�25
(C) 7.9 x 10�6
(D) 1.58 x 10�5
14. The mass of a nitrogen gas molecule is 14 times that of a hydrogen molecule. What
is the hydrogen molecule's velocity, relative to that of the nitrogen molecule (VN2),
if both are at the same temperature?
(A) 0.27 VN2
(B) 3.7 VN2
(C) 14 VN2
(D) 196 VN2
15. The Ksp of PbBr2 is 6.3 x 10�6. If 50 mL of 0.020
M Pb(NO3)2 are mixed with 50 mL of 0.010 M CaBr2, which
of the following is true?
(A) the solution will not form a precipitate
(B) calcium nitrate will precipitate
(C) PbBr2 will precipitate and excess Pb2+ will remain in solution
(D) PbBr2 will precipitate and excess Br� will remain in solution
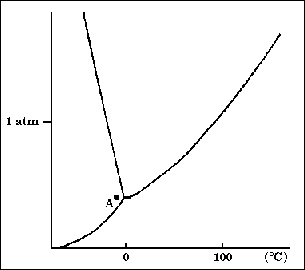
16. According to the phase diagram to the right, if the pressure is increased at
constant temperature, from point A, what change will occur?
(A) Solid will change to vapor
(B) Liquid will change to vapor
(C) Liquid will change to solid
(D) Solid will change to liquid
17. How many of the following salts will be more soluble in acid solution than in pure
water?
CdCO3, Mn(OH)2, PbS, PbCl2
(A) 1
(B) 2
(C) 3
(D) 4
18. A 50.0 mL sample of nitrogen gas is collected over water at 25 �C and 720 torr.
What is the volume in mL of the dry gas at STP? (The vapor pressure of water at 25 �C is
23.8 torr.)
(A) 42.0
(B) 43.4
(C) 57.6
(D) 59.6
19. To which of the following would addition of an equal volume of 0.50 M NaOH lead to
a solution having a lower pH?
(A) 0.30 M HCl
(B) water
(C) 0.60 M KOH
(D) 0.40 M NaNO3
20. Consider the solvent effect on acid dissociation. Would you expect HCl to be weak
or strong in the two solvents, water and benzene (C6H6)?
(A) strong in both
(B) weak in both
(C) strong in water, weak in benzene
(D) weak in water, strong in benzene
21. The equilibrium constant for the equation below is very small; that is, K <<
1.
HX (aq) + Y�(aq) <===> X� (aq) + HY (aq)
Which statement is true?
(A) The reaction goes nearly to completion
(B) HY is a weaker acid than HX
(C) X� is a better proton acceptor than Y�
(D) Y� is a stronger base than X�
22. Consider the reaction 3A (g) + 2B (g) <===> C (g). Initially, only A and C
are present, each at a concentration of 0.20 M. At equillibrium, the concentration of B is
0.10 M. What is the value of the equilibrium constant, Kc, for the reaction?
(A) 6
(B) 43
(C) 60
(D) 350
23. Consider the system
NH4Cl (s) <===> NH3 (g) + HCl (g)
If the concentration of ammonia gas is tripled, the value of the equilibrium constant
will
(A) triple
(B) increase, buy by more than a factor of three
(C) decrease to one-third its value
(D) remain the same
24. The weak base ionization constant (Kb) for hydroxylamine, HONH2,
is 1.1 x 10�8. Which of the following equations best describes its ionization
equilibrium?
(A) HONH2 (aq) + H2O (l) ---> HONH3+
(aq) + OH� (aq)
(B) HONH2 (aq) ---> NH2+ (aq) + OH� (aq)
(C) HONH2 (aq) + H2O (l) ---> ONH2� (aq) + H3O+
(aq)
(D) HONH2 (aq) + H3O+ (aq) ---> HONH3+
(aq) + H2O (l)
25. Which substance, when added to water, will NOT change the pH?
(A) NaHCO3
(B) NH4Cl
(C) KCN
(D) KCl
26. How many O2 molecules are contained in 2.0 liters of oxygen gas at 27
�C and 3.0 atm pressure?
(A) 1000
(B) 1.5 x 1023
(C) 1.5 x 1024
(D) 2.5 x 1024
27. What is the vapor pressure of an isopropanol solution containing 80.0 g of the
nonvolatile solute glycerol (C3H8O3, molar mass = 92.0 g
mol�1) and 212 g is isopropanol (C3H8O, molar mass =
60.0 g mol�1) at 68 �C? (The vapor pressure of isopropanol at 68 �C is 400
torr.)
(A) 79
(B) 212
(C) 321
(D) 400
28. If 40.0 mL of 0.20 M CH3COOH are titrated with 0.20 M NaOH, how many mL
of base must be added to form a buffer solution with the greatest buffering capacity?
(A) 5.0
(B) 10.0
(C) 20.0
(D) 40.0
29. An important reaction in the production for nitrogen fertilizers is
4NH3 (g) + 5O2 (g) --> 4NO (g) + 6 H2O
(g)
At constant temperature and pressure, what is the maximum number of liters of NO (g)
that can be produced from 14.0 L of NH3 and 16.0 L of O2?
(A) 12.8
(B) 14.0
(C) 17.5
(D) 20.0
30. Which of the following graphs for an ideal gas, when all other variables are held
constant, will NOT yield a straight line?
(A) P versus 1/V
(B) V versus T
(C) n versus T
(D) P versus T
31. What is the equilibrium constant for a reaction that has a value of DG = - 41.8 kJ at 100 �C?
(A) -5.87
(B) 1.4 x 10�6
(C) 13.5
(D) 7.1 x 105
32. How much heat must be removed from a 10.0 g ingot of stainless steel in order to
lower its temperature from 60.0 �C to 35.0 �C? (The specific heat of stainless steel is
0.51 J g�1 K�1.)
(A) 128 J
(B) 179 J
(C) 306 J
(D) 409 J
33. What is the standard molar entropy change, DS,
for the synthesis of ammonia, using the following data.
| |
1/2 N2 (g) |
+ |
3/2 H2 (g) |
---> |
NH3 (g) |
| S� (J K mol�1) |
192 |
|
131 |
|
193 |
(A) -99.5 J K�11
(B) -130 J K�1
(C) +130 J K�1
(D) +516 J K�1
34. Which of the following reactions is accompanied by a decrease in entropy?
(A) H2O (l) ---> H2O (g)
(B) C2H2 (g) + 2 H2 (g) ---> C2H6
(g)
(C) NH4Cl (s) + H2O ---> NH4Cl (aq)
(D) H2 (g) + Cl2 ---> 2 HCl
35. Trouton's Rule, which states that font face="Symbol">DHvap
/ Tbp = 85 J mol�1 K�1 (a constant) for nonpolar
liquids, relates to the entropy change for the vaporization process, DSvap.
Knowing this, how would the value for water differ from this constant?
(A) It would be smaller than 85 J mol�1 K�1.
(B) It would be larger than 85 J mol�1 K�1.
(C) It would also be 85 J mol�1 K�1.
(D) It is impossible to predict.
36. At equilibrium, the Gibbs free energy function of a reaction, DG,
has a value
(A) greater than zero.
(B) less than zero.
(C) equal to zero.
(D) which depends on the particular system.
37. An ammonia solution has a density of 0.910 g cm�3 and is 25.0% NH3
by mass. What is the molarity of the solution?
(A) 12.1 M
(B) 13.4 M
(C) 14.5 M
(D) 15.5 M
38. Which of the following molecules has a measurable dipole moment?
(A) BrF3
(B) XeF4
(C) CF4
(D) PCl5
39. Which type of crystal structure best describes that present in diamond?
(A) ionic
(B) covalent
(C) metallic
(D) extended molecular
40. Which of the following substances has the highest melting point?
(A) KCl
(B) ClO2
(C) CaO
(D) BiCl3
41. Which of the following does not define a covalent bond?
(A) a shared pair of electrons
(B) an overlap of half-filled atomic or hybrid orbitals
(C) increased electron density in the region between atoms of two nonmetals
(D) electrostatic attraction between species in which one or more electrons have been
transferred
42. How many equivalent contributing resonance forms does the ion NO3�
have?
(A) 1
(B) 2
(C) 3
(D) 4
43. Which compound is the anhydride of sulfurous acid?
(A) SO4
(B) SO3
(C) SO2
(D) Na2SO4
44. Which is the most accurate description of Na+ species in dilute aqueous
solution?
(A) the nearest neighbor is a Cl� ion
(B) the nearest neighbor is the positive end of the water dipole
(C) the nearest neighbor is the negative end of the water dipole
(D) there are no interactions with any species in the solution
45. Which of the following oxides, at the same concentration when dissolved in water,
results in the most acidic solution?
(A) CO2
(B) B2O3
(C) N2O5
(D) Li2O2
46. Which of the following salts should form a colorless solution when dissolved in
water?
(A) CuCl2
(B) FeCl3
(C) CrCl3
(D) CaCl2
47. Which of the following can act as a Lewis base?
(A) BF3
(B) CH4
(C) PH3
(D) NH4+
48. The shape of the phosphite ion, PO33�, is best described as
(A) tetrahedral.
(B) trigonal planar.
(C) trigonal pyramidal.
(D) bent.
49. What is the ground state electron configuration of the Mn2+ ion?
(A) [Ar] 4s1 3d5
(B) [Ar] 4s2 3d3
(C) [Ar] 3d5
(D) [Ar] 3d4
50. Which group of metals has the lowest melting points?
(A) alkali metals
(B) alkaline earth metals
(C) transition metals
(D) lanthanide metals
51. Which solution has the highest electrical conductivity?
(A) 0.05 M KCl
(B) 0.05 M HF
(C) 0.05 M NH3
(D) 0.05 M CaCl2
52. Which of the following compounds exhibit geometric isomerism?
| I. |
CH3CH=CHCH2CH3 |
| II. |
CH2=CHCl |
| III. |
CH3CH=CHCl |
(A) I and III
(B) I and II
(C) II and III
(D) I, II, and III
53. What hybrid orbitals are used by each nitrogen atom in N2F2?
(A) sp
(B) sp2
(C) sp3
(D) p only
54. From the Lewis structures given below, choose the invalid one.
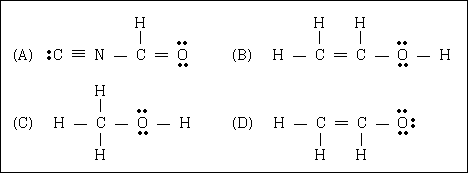
55. The compounds Br2 (159.8 g mol�1) and ICl (162.4 g mol�1)
have similar molar masses, yet ICl boils at 97 �C and Br2 boils at 59 �C.
Which statement best explains this difference?
(A) ICl has a longer bond that Br2.
(B) ICl is ionic, while Br2 is covalently bonded.
(C) ICl has a measurable dipole movement, but Br2 does not.
(D) ICl has a stronger bond than Br2.
56. Which one of the following salts is expected to give a red color in a flame test?
(A) KCl
(B) NaCl
(C) CaCl2
(D) BaCl2
57. Which of the following metals exhibits the greatest variety of oxidation states?
(A) Ba
(B) Y
(C) Cr
(D) Tl
58. In spontaneous beta particle (b�) emission, what is the
source of the emitted electron?
(A) the nucleus
(B) the 1s orbital
(C) the outermost occupied orbital
(D) a random orbital
59. Which one of the compounds below is classified as a peroxide?
(A) MgO
(B) Na2O
(C) BaO2
(D) KO2
60. Which of the following sets of ions is generally associated with "hard"
water?
(A) Na+, Ca2+
(B) Na+, K+
(C) Ca2+, Mg2+
(D) Mg2+, SO42�
61. The boiling points of four liquids are shown. Which liquid has the
highest vapor pressure at 25 �C?
| Compound |
Boiling Point |
| ethyl ether |
35 �C |
| acetone |
56 �C |
| ethanol |
78 �C |
| water |
100 �C |
(A) ethyl ether
(B) acetone
(C) ethanol
(D) water
62. Which term best describes a 0.10 M solution of Fe(NO3)3?
(A) saturated
(B) neutral
(C) acidic
(D) basic
63. Very strong acids, such as HNO3 and HCl, appear to be equally strong in
water. This "leveling effect" of water occurs because
(A) OH� is a stronger base than the conjugate bases of HNO3 and HCl.
(B) H3O+ is a stronger acid than HNO3 and HCl.
(C) H2O is a base than the conjugate bases of HNO3 and HCl.
(D) H2O is a weaker base than the conjugate bases of HNO3 and HCl.
64. If the equilibrium P4(g) + 6 Cl2(g) <===> 4 PCl3(g)
is established by adding equal numbers of moles of P4 and Cl2 to an
evacuated flask, which of the following must be true at equilibrium?
(A) [Cl2] > [PCl3]
(B) [Cl2] < [P4]
(C) [PCl3] > [P4]
(D) [P4] = [PCl3]
65. Consider the equilibrium NH4HS(s) <===> NH3(g) + H2S(g).
A 4.65 g sample of solid NH4HS is placed in an evacuated 3.0 L flask at 35 �C
and allowed to reach equilibrium at which time the total pressure in the flask is 0.82
atm. What is the value of Kp at this temperature?
(A) 0.67
(B) 0.41
(C) 0.17
(D) 0.45
66. When propanol, CH3CH2CH2OH, is oxidized to
propanoic acid, CH3CH2COOH, what is the number of electrons on the
right side of the half-reaction?
(A) 2
(B) 3
(C) 4
(D) 5
67. What is the [Cu2+] in the cell Zn / Zn2+ (0.05 M) // Cu2+
(X M) / Cu if the cell voltage is 1.03 V?
(A) 0.12 M
(B) 0.0002 M
(C) 0.05 M
(D) 0.0035 M
68. Ten amperes are passed through molten aluminum chloride for 5.5 hours. How many
grams of aluminum metal could be produced by this electrolysis?
(A) 18.5
(B) 55.4
(C) 91.2
(D) 273
69. What is the DG� for the reaction 2 Al3+ + 6
I�---> 2 Al + 3 I2, at 298 K?
(A) +1300 kJ
(B) -650 kJ
(C) -420 kJ
(D) 650 kJ
70. How many of the following would shift this reaction to the right?
PbO2(s) + 4 H+ + Cu(s) ---> Pb2++ 2
H2O + Cu2+
Acid is added.
PbO2 is added.
The [Pb2+] is increased.
The pH is increased.
Sulfide ion is added.
(A) 1
(B) 2
(C) 3
(D) 4
|