U.S. National Chemistry Olympiad: 1991 Local
Section Test
Go to answers
1. What is the maximum amount of MgCl2 that can be prepared from the
reaction of 10.0g of HCl with 10.0 of Mg(OH)2 ?
2 HCl + Mg(OH)2 ---> MgCl2 + 2 H2O
(A) 9.26 g
(B) 13.1 g
(C) 16.3 g
(D) 17.5 g
2. Phenylflouroform contains 57.54% C, 3.45% H, and 39.01% F. Its molecular formula is
the same as its simplest formula. How many carbon atoms are in one molecule of
Phenyflouroform ?
(A) 4
(B) 5
(C) 6
(D) 7
3. What is the most likely formula for a compound of gallium and sulfur ?
(A) GaS
(B) GaS2
(C) Ga2S3
(D) Ga3S2
4. A mixture of 2.30 x 1023 molecules N2 and 4.35 x 1023
molecules N2O has a total pressure of 2.00 atm. What is the partial pressure of
N2 ?
(A) 0.346 atm
(B) 0.654 atm
(C) 0.692 atm
(D) 1.11 atm
5. Which element should have the smallest first ionization energy ?
(A) Ca
(B) Rb
(C) Al
(D) Cl
6. An aqueous solution containing 1.00 mol H2SO4 is mixed with an
aqueous solution containing 1.00 mol NaOH. The mixture is then evaporated to dryness. What
solid remains after evaporation ?
(A) H2SO4
(B) NaOH
(C) NaHSO4
(D) Na2SO4
7. Which atom is the smallest ?
(A) Rb
(B) Ag
(C) Sb
(D) I
8. At room temperature and 1 atm pressure the molecules are farthest apart in
(A) flourine
(B) bromine
(C) iodine
(D) water
9. What is the net-ionic equation for the reaction of aqueous solutions of BaCl2
and K2SO4?
(A) Ba2+(aq) + 2ClŻ(aq) + 2K+(aq) + SO42Ż(aq)
---> BaSO4(s) + 2K+(aq) + 2ClŻ(aq)
(B) Ba2+(aq) + SO42Ż(aq) ---> BaSO4(s)
(C) K+(aq) + ClŻ(aq) ---> KCl(s)
(D) Ba2+(aq) + 2ClŻ(aq) + 2K+(aq) + SO42Ż(aq)
---> BaSO4(s) + 2KCl(s)
10. When 80.00 mL of 0.200 M HNO3 is added to 120.00 mL of 0.150 M KOH, the
reaction
HNO3(aq) + KOH(aq) ---> KNO3(aq) + H2O(l)
occurs and the resulting solution is
(A) 0.0160 M KNO3
(B) 0.0180 M KNO3
(C) 0.0800 M KNO3 and 0.0100 M KOH
(D) 0.0160 M KNO3 and 0.0200 M KOH
11. Methanol burns in oxygen according to the equation
CH3OH + O2 ---> CO2 + H2O
(unbalanced)
What volume of oxygen is required to burn 5.0 L of gaseous methonal measured at the
same temperature and pressure ?
(A) 15.0 L
(B) 3.3 L
(C) 7.5 L
(D) 5.0 L
12. A 50 g sample of an ideal gas occupies a volume of 3.2 L at 40 °C and exerts a
pressure of 2.6 atm. What is its molar mass ?
(A) 150 g molŻ1
(B) 2.0 g molŻ1
(C) 77 g molŻ1
(D) 310 g molŻ1
13. When sodium metal reacts with an excess of water the products are
(A) Na2O(s) and H2(g)
(B) Na+(aq), OHŻ(aq) and H2(g)
(C) NaH(s) and O2(g)
(D) Na2O2(s) and H2(g)
14. Which is least important in determining the physical behavior of a gas?
(A) volume
(B) pressure
(C) temperature
(D) chemical composition
15. Which pair of atoms should form the most polar bond ?
(A) B and F
(B) C and O
(C) O and F
(D) F and N
16. Which pair of ions should form the strongest ionic bond?
(A) Li+ and IŻ
(B) Na+ and FŻ
(C) Cs+ and FŻ
(D) Na+ and O2Ż
17. The kinetic-molecular theory predicts that two gases at the same temperature will
have the same
(A) average speed
(B) average kinetic energy
(C) pressure
(D) rate of effusion
18. Which factors do not affect the vapor pressure of a liquid at equilibrium?
I. Intermolecular forces of attraction.
II. The volume of liquid present.
III. The temperature of the liquid
(A) I only
(B) II only
(C) I and II only
(D) II and III only
19. Which has the highest vapor pressure at room temperature ?
(A) a vegetable oil
(B) gasoline
(C) table sugar, C12H22O11
(D) table salt, NaCl
20. Identify the best Lewis structure for hydrogen cyanide.
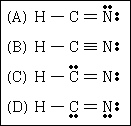
21. Which oxide of chlorine exhibits the maximum oxidation state that really exists for
chlorine?
(A) ClO
(B) ClO4
(C) Cl2O5
(D) Cl2O7
22. Which has the largest radius ?
(A) Na
(B) Na+
(C) FŻ
(D) Ne
23. What is the molarity of HCl if 450 mL of 2.3 M HCl is diluted to 1250 mL ?
(A) 0.36 M
(B) 0.50 M
(C) 0.83 M
(D) 1.8 M
24. The first five ionization energies in kJ molŻ1, for a particular
element are shown.
| 1st |
2nd |
3rd |
4th |
5th |
| 580 |
1815 |
2740 |
11,600 |
18,500 |
The element is likely to form a ionic compounds in which its charge is
(A) 1+
(B) 2+
(C) 3+
(D) 4+
25. Which compound, when dissolved in water, conducts electricity and forms a basic
solution ?
(A) HCl
(B) CH3COOH
(C) CH3OH
(D) KOH
26. Which pair constitutes a buffer ?
(A) HCl and KCl
(B) NaOH and NaCl
(C) HNO2 and NaNO2
(D) HNO3 and NH4NO3
27. Which oxide dissolves in water to give the strongest acid ?
(A) SO3
(B) CaO
(C) P4O10
(D) CO2
28. Which is the strongest Bronsted base ?
(A) NH2Ż
(B) ClŻ
(C) PO43Ż
(D) NO2Ż
29. In the reaction FŻ + H2O ---> HF + OHŻ an acid-base conjugate pair
is
(A) FŻ and H2O
(B) FŻ and OHŻ
(C) H2O and HF
(D) FŻ and HF
30. Which is the anhydride of nitric acid ?
(A) NO
(B) NO2
(C) N2O3
(D) N2O5
31. Which forms a basic aqeous solution ?
(A) NaI
(B) KNO3
(C) BaCl2
(D) Na2CO3
32. The best acid for preparing a buffer of pH = 3.2 has a Ka near
(A) 2.0 x 10Ż5
(B) 6.0 x 10Ż4
(C) 3.2 x 10Ż2
(D) 4.0 x 10Ż6
33. What is the pH of a 2.0 x 10Ż3 M HCl solution ?
(A) 2.40
(B) 2.70
(C) 3.00
(D) 11.30
34. Consider a solution that contains only one ion, Ag+, Pb2+, or
Hg22+. The solution gave a precipitate with HCl that was insoluble
in hot water but was soluble in dilute ammonia solution. Which ion was present ?
(A) Pb2+
(B) Ag+
(C) Hg22+
(D) The information is insufficient to identify the ion
35. What is the partial pressure of CO2(g) if Kp for the reaction
is 1.04 atm ?
CaCO3(s) <===> CaO(s) + CO2(g)
(A) 0.52 atm
(B) 1.04 atm
(C) 1.08 atm
(D) 10.4 atm
36. The Ksp values for CaSO4,BaSO4, and Ag2SO4
are 2.0 x 10Ż4, 1.5 x 10Ż9, and 1.5 x 10Ż5
respectively. If 0.010 M Na2SO4 is slowly added to a solution that
contains Ca2+, Ba2+, and Ag+ (each 0.10M), which solid
will precipitate last?
(A) CaSO4
(B) Ag2SO4
(C) BaSO4
(D) More information is required
37. The Ksp of AgCl is 1.8 x 10Ż10. If 10.00 mL of 0.0010 M NaCl
is combined with 20.00 mL of 0.0050 M AgNO3
(A) no reaction occurs
(B) a precipitate forms
(C) an initial precipitate forms which dissolves upon stirring.
(D) the silver ion concentration remainsas 0.005 M.
38. N2(g) + 3H2(g) <===> 2NH3(g) + 92.2 kJ
The partial pressure of N2 is not increased by
(A) decreasing container size
(B) adding NH3
(C) increasing temperature
(D) removing H2
39. Which value for an equilibrium constant, K, could indicate that the reaction
A(g) + B(g) ---> C(g)
goes nearly to completion?
(A) 1014
(B) 10Ż14
(C) 1
(D) 0
40. Which is most soluble in a polar solvent?
(A) CF4
(B) Br2
(C) HCl
(D) BCl3
41. When a liquid is placed in a closed container
(A) evaporation stops
(B) evaporation continues for a time then stops
(C) the vapor pressure above the liquid becomes one atmosphere
(D) evaporation and condensation continue to occur, but at equal rates
42. The sublimination of dry ice is an endothermic process. The energy absorbed is
required to overcome
(A) covalent bonds
(B) polar covalent bonds
(C) dipole forces
(D) dispersion forces
43. Compared to a 1.0 M aqueous solution of a nonelectrolyte, a 1.0 M solution of an
electrolyte will have a
(A) higher melting point and higher boiling point
(B) higher melting point and lower boiling point
(C) lower melting point and lower boiling point
(D) lower melting point and higher boiling point
44. Which is planar?
(A) H3O+
(B) PCl3
(C) CO32Ż
(D) NH4+
45. Calculate DH°f for
4 NH3(g) + 3 O2(g) ---> 2 N2(g) + 6
H2O(g)
Given that DH°f for NH3(g) and H2O(g)
are -46.0 and -242 kJ molŻ1 respectively.
(A) -1268 kJ
(B) -196 kJ
(C) +1268 kJ
(D) +196 kJ
46. A white, odorless, crystalline solid melts after about ten seconds in a burner
flame. It is soluble in water and insoluble in carbon tetrachloride. Neither the melt nor
its aqueous solution conducts electricity. We conclude that the substance is
(A) an ionic compound
(B) a non-polar covalent compound
(C) a polar covalent compound
(D) a pure element
47. Given the standard reduction potentials,
| Cr3+ + 3eŻ ---> Cr |
-0.74 V |
| Pb2+ + 2eŻ ---> Pb |
-0.13 V |
what is the standard potential, E°, for the following reaction?
2 Cr + 3 Pb2+ ---> 2 Cr3+ + 3 Pb
(A) 0.61 V
(B) 0.87 V
(C) 1.09 V
(D) 1.87 V
48. A mixture of 0.10 mol H2 and 0.050 mol O2 are placed in a
bomb calorimeter with a heat capacity of 5.1 x 104 J °CŻ1. The
initial temperature is 25.00 °C and the temperature after combustion is 25.56 °C. What
is the DH°f for H2O(l)? (Assume 100%
conversion to water.)
(A) -286 kJ molŻ1
(B) +190 kJ molŻ1
(C) -190 kJ molŻ1
(D) +286 kJ molŻ1
49. In the reaction
2MnO4Ż + 16H+ + 10BrŻ ---> 2Mn2+ +
8H2O + 5Br2
BrŻ ion is the
(A) oxidizing agent and is oxidized
(B) oxidizing agent and is reduced
(C) reducing agent and is oxidized
(D) reducing agent and is reduced
50. For BrF3, the orbital geometry of the central atom and the molecular
geometry are, respectively,
(A) trigonal planar and trigonal planar
(B) trigonal planar and trigonal bipyramidal
(C) trigonal bipyramidal and trigonal planar
(D) trigonal bipyramidal and T-shaped
51. How many electrons are needed to convert one nitrate ion into one ammonia molecule?
(A) 3
(B) 5
(C) 6
(D) 8
52. Which species below violates the octet rule?
(A) CF4
(B) BF3
(C) N2O
(D) NO3Ż
53. What is the coefficient for H2O when the redox equation
Cu + NO3Ż + H+ ---> Cu2+ + NO + H2O
is balanced using smallest whole - number coefficients?
(A) 1
(B) 2
(C) 3
(D) 4
54. Which change will produce an increase in the entropy of a system?
(A) decreasing the temperature
(B) forming a precipitate in a solution
(C) decreasing the volume
(D) forming a gas from a solid reaction
55. Which will cause the value of the equilibrium constant for this reaction to
increase?
2A(g) + B(g) ---> C(g); DH° = 280 kJ
(A) adding a catalyst
(B) increasing the concentration of A
(C) increasing the concentration of C
(D) increasing the temperature
56. Given
2CO(g) + O2(g) ---> 2CO2(g); DG° = -516 kJ
4MnO(s) + O2(g) ---> 2Mn2O3(s); DG° = -312 kJ
Calculate the value of DG° for
2MnO(s) + CO2(g) ---> Mn2O3(s) +
CO(g)
(A) +102 kJ
(B) -102 kJ
(C) +204 kJ
(D) -204 kJ
57. Given
Br2(l) S° = 152.2 J molŻ1 KŻ1 and DH°f = 0 kJ molŻ1
Br2(g) S° = 245.4 J molŻ1 KŻ1 and DH°f
= 30.9 kJ molŻ1
Calculate the boiling point of liquid bromine.
(A) 58.4 K
(B) 332 K
(C) 125 K
(D) -147 K
58. Which is not expected to have a value of zero?
(A) DH°f for Cl2(g)
(B) DG°f for Na(s)
(C) S° for Ca(s)
(D) DH°f for Al(s)
59. The half-life of 14C is 5570 years. How many years will it take for 90%
of a sample to decompose?
(A) 5,570 years
(B) 17,700 years
(C) 18,600 years
(D) 50,100 years
60. A solution containg 2.10 g of Fe(NH4)2(SO4)2
. 6 H2O (M = 392.1 g molŻ1) was titrated with
acidic Na2Cr2O7 solution, requiring 41.60 mL of Na2Cr2O7
solution. What is the molarity of Na2Cr2O7?
6 Fe2+ + Cr2O72Ż + 14 H+
---> 6 Fe3+ + 2 Cr3+ + 7 H2O
(A) 0.0215 M
(B) 0.0387 M
(C) 0.0644 M
(D) 0.0744 M
61. What is the approximate H-O-H bond angle in H3O+?
(A) 60
(B) 90
(C) 109
(D) 120
62. What hybrid orbitals are used by nitrogen in N2F2?
(A) sp
(B) sp2
(C) sp3
(D) p only
63. The strongest interaction between atoms listed below is
(A) an instantaneous dipole-dipole interaction
(B) a dipole-dipole interaction
(C) a hydrogen bond
(D) a covalent bond
64. When a 5.0 g sample of an unknown compound is dissolved in 250.0 g of benzene, the
freezing point of benzene decreased 1.4 °C. The freezing point depression constant, Kf,
for benzene is 5.12 °C mŻ1. What is the approximate molar mass of the
compound?
(A) 290 g molŻ1
(B) 73 g molŻ1
(C) 36 g molŻ1
(D) 140 g molŻ1
65.
| Half-cell reaction |
Standard reduction potential, E° |
| Cu2+ (aq) + 2eŻ ---> Cu(s) |
-0.25 V |
| Ni2+ (aq) + 2eŻ ---> Ni(s) |
+0.34 V |
When two half-cells are connected using a salt bridge,
(A) a galvanic cell will result in which Cu is the cathode
(B) a galvanic cell will result in which Cu is the anode
(C) an electrolytic cell will result in which Ni is the anode
(D) an electrolytic cell will result in which Ni is the anode
66. What is the maximum number of electrons that can occupy the 5d subshell?
(A) 2
(B) 6
(C) 10
(D) 14
67. For the isoelectronic series below, which species requires the least energy to
remove an outer electron?
(A) K+
(B) Ar
(C) ClŻ
(D) S2Ż
68. In aqueous solution, iodide ion (in basic solution) is oxidized by hypochlorite
ion:
OClŻ(aq) + IŻ(aq) ---> OIŻ(aq) + ClŻ(aq)
The rate of formation of hypoiodite, OIŻ, is given by the rate law
rate = ( k [OClŻ] [IŻ] ) / [OHŻ]
What is the overall reaction order for the formation of OIŻ?
(A) 1
(B) 2
(C) 3
(D) 4
69. The dissociation of HI molecules, as shown below, occurs at a temperature of 629 K.
The rate constant, k = 3.02 x 10Ż5 MŻ1 sŻ1.
2 HI(g) ---> H2(g) + I2(g)
What is the reaction order?
(A) 0
(B) 1
(C) 2
(D) 3
70. A sodium vapor street lamp emits yellow light with a wavelength of 589 nm. What is
the energy of this light per mole of protons?
(A) 3.37 x 10Ż16 kJ molŻ1
(B) 2.35 x 10Ż13 kJ molŻ1
(C) 0.201 kJ molŻ1
(D) 203 kJ molŻ1
|